Unveiling the Mysteries of EDTA
I. Introduction
In the modern world, we are constantly surrounded by various chemicals, some of which are so deeply ingrained in our daily lives that we may not even be aware of their presence. One such chemical is Ethylenediaminetetraacetic acid, commonly known as EDTA. From the food we eat to the cleaning products we use and the skincare items we apply, EDTA seems to be everywhere. But the question remains: Is EDTA good or bad for you? Why do they put EDTA in food? Is EDTA safe for skin? And what is EDTA used for in cleaning? This article aims to explore these questions in depth, providing a comprehensive understanding of this versatile compound.
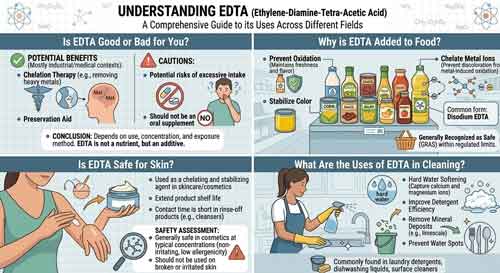
II. Is EDTA Good or Bad for You?
1. Chelating Action
EDTA is a chelating agent, which means it has the unique ability to bind to metal ions. In the human body, it can form stable complexes with metal ions such as calcium, magnesium, lead, mercury, and iron. In some medical applications, this property is harnessed for chelation therapy. For example, in cases of heavy metal poisoning, EDTA can be used to remove toxic metals from the body. It binds to the metal ions, forming a complex that can then be excreted through the kidneys.
2. Nutrient Depletion Concerns
However, this chelating action also raises concerns. Since EDTA can bind to essential metal ions like calcium and magnesium, long - term or excessive use could potentially lead to a depletion of these nutrients in the body. Calcium is vital for bone health, muscle function, and nerve transmission, while magnesium is involved in over 300 enzymatic reactions in the body, including energy production and DNA synthesis. If EDTA is present in high enough amounts in the body, it may disrupt these normal physiological processes by sequestering these essential minerals.
3. Safety in Moderation
In general, when used appropriately, the benefits of EDTA in medical settings often outweigh the potential risks. The dosage and duration of EDTA treatment are carefully monitored by medical professionals to ensure that the removal of toxic metals is maximized while minimizing the impact on essential nutrients. For the general population, the low levels of EDTA present in food and consumer products are considered safe, as the body's natural regulatory mechanisms can handle the small amounts of EDTA that may enter the system.
III. Why Do They Put EDTA in Food?
1. Preventing Oxidation
One of the main reasons EDTA is added to food is its ability to act as a preservative and antioxidant. Many foods contain metal ions, such as iron and copper, which can catalyze oxidation reactions. Oxidation can lead to the spoilage of food, causing changes in color, flavor, and nutritional value. EDTA binds to these metal ions, preventing them from participating in oxidation reactions. For example, in salad dressings, EDTA helps to prevent the oxidation of fats, which would otherwise lead to rancidity.
2. Enhancing Shelf - Life
By inhibiting oxidation, EDTA significantly extends the shelf - life of many food products. This is especially important for processed foods that need to be stored for long periods before consumption. In canned fruits and vegetables, EDTA can prevent the discoloration caused by the reaction of metal ions with phenolic compounds in the food. It also helps to maintain the texture and nutritional quality of the food over time, reducing food waste.
3. Meeting Regulatory Requirements
Food manufacturers add EDTA to their products in accordance with strict regulatory guidelines. Regulatory bodies around the world, such as the Food and Drug Administration (FDA) in the United States, have established acceptable daily intake (ADI) levels for EDTA in food. These limits ensure that the amounts of EDTA added to food are safe for human consumption. As long as manufacturers adhere to these regulations, the use of EDTA in food is considered a valuable tool for maintaining food quality and safety.
IV. Is EDTA Safe for Skin?
1. Limited Skin Penetration
EDTA has relatively low skin penetration. The outer layer of the skin, the stratum corneum, acts as a natural barrier that prevents many substances from entering the body. When applied topically, only a small fraction of EDTA is able to penetrate the skin. This limited penetration reduces the risk of systemic effects from skin exposure.
2. Allergic Reactions
However, like any substance, some individuals may be allergic to EDTA. Allergic reactions to EDTA can range from mild skin irritations, such as redness, itching, and swelling, to more severe reactions in rare cases. These allergic reactions are usually due to the body's immune system recognizing EDTA as a foreign substance and mounting an immune response. People with sensitive skin or a history of allergies may be more prone to developing such reactions.
3. Use in Skincare Products
Despite the potential for allergic reactions, EDTA is widely used in skincare products. In these formulations, it serves multiple functions. It can enhance the effectiveness of other ingredients by preventing the inactivation of preservatives by metal ions. It also helps to maintain the stability of the product, ensuring that the active ingredients remain in their proper form over time. Manufacturers are required to conduct safety assessments to ensure that the concentration of EDTA in skincare products is within safe limits and to label their products appropriately, so consumers can make informed decisions.
V. What is EDTA Used for in Cleaning?
1. Water Softening
One of the primary uses of EDTA in cleaning is water softening. Hard water contains high levels of calcium and magnesium ions, which can cause problems in cleaning processes. These metal ions can react with soap and detergents, forming insoluble precipitates that reduce the cleaning effectiveness. EDTA binds to these calcium and magnesium ions, effectively softening the water. This allows soap and detergents to lather better and work more efficiently, resulting in cleaner surfaces.
2. Detergent Booster
In addition to water softening, EDTA acts as a detergent booster. It helps to break down dirt and grease by preventing the redeposition of soil particles onto the cleaned surface. EDTA binds to metal ions present in the dirt, which can otherwise cause the dirt to adhere to the surface. By sequestering these metal ions, EDTA enhances the cleaning power of detergents, making them more effective at removing tough stains.
3. Cleaning Different Surfaces
EDTA is used in a wide range of cleaning products, from laundry detergents to household cleaners for countertops, floors, and bathrooms. In laundry, it helps to prevent the buildup of mineral deposits on clothes, keeping them looking brighter and feeling softer. In household cleaners, it can effectively remove stains caused by hard water, rust, and other mineral - based contaminants. It is also used in industrial cleaning applications, such as cleaning boilers and pipes, where the removal of scale and mineral deposits is essential for maintaining equipment efficiency.
VI. EDTA in Other Applications
1. Stabilizing Medications
In the pharmaceutical industry, EDTA is used to stabilize medications. Many drugs contain metal - sensitive components, and the presence of metal ions can cause degradation of the drug over time. EDTA binds to these metal ions, preventing them from reacting with the drug and thus extending the shelf - life of the medication. It is commonly used in injectable medications, eye drops, and oral liquid formulations.
2. Enhancing Drug Delivery
EDTA can also enhance drug delivery in some cases. By binding to metal ions in the body, it can modify the local environment around the drug, potentially improving its absorption and distribution. For example, in some topical medications, EDTA can help the drug penetrate the skin more effectively by altering the skin's permeability.
3. Preservative and Formula Stabilizer
Similar to skincare products, EDTA is used in cosmetics for its preservative and formula - stabilizing properties. It helps to prevent the growth of microorganisms in cosmetics, which can lead to product spoilage and potential harm to the user. By binding to metal ions, it also ensures the stability of the cosmetic formulation, preventing separation, discoloration, and changes in texture.
VII. Regulatory Oversight of EDTA
1. Food Regulations
As mentioned earlier, regulatory bodies worldwide have established guidelines for the use of EDTA in food. In the European Union, the use of EDTA in food is regulated by specific directives that define the permitted levels and food categories in which it can be used. The Joint FAO/WHO Expert Committee on Food Additives (JECFA) has also set an acceptable daily intake for EDTA, which serves as a reference for countries in establishing their own regulations.
2. Cosmetics and Cleaning Product Regulations
For cosmetics and cleaning products, regulations also govern the use of EDTA. In the United States, the FDA regulates the use of EDTA in cosmetics to ensure their safety. In the cleaning product industry, regulations ensure that the concentrations of EDTA used are safe for consumers and the environment. These regulations often require manufacturers to conduct safety testing and label their products accurately.
VIII. Conclusion
In conclusion, EDTA is a complex and versatile compound with a wide range of applications in our daily lives. When it comes to the question of whether EDTA is good or bad for you, the answer lies in the context of its use. In medical settings, it can be a life - saving treatment for heavy metal poisoning, but excessive or improper use may lead to nutrient depletion. In food, it plays a crucial role in preservation and maintaining food quality, and when used within regulatory limits, it is safe for consumption. Regarding skin safety, while most people can use products containing EDTA without issue, a small percentage may be allergic. In cleaning, EDTA is an effective water - softening and detergent - boosting agent. Overall, with proper regulatory oversight and understanding of its properties, EDTA can continue to be a valuable tool in various industries, enhancing our health, food safety, and cleaning efficiency. As consumers, being informed about the presence and uses of EDTA in the products we use allows us to make more educated choices about our health and well - being.
